Clinical trials are among the most critical elements of the drug development process, but they are also one of the most complex. Study designs are more elaborate than ever and are often held across multiple sites, spanning many countries, each with its own set of unique requirements.
Agile and efficient supply chains are required to effectively serve globally dispersed sites, delivering costly comparator drugs and delicate investigative compounds. This process involves paying close attention to a wide range of factors, including inventory, demand planning, compliance, and logistics.
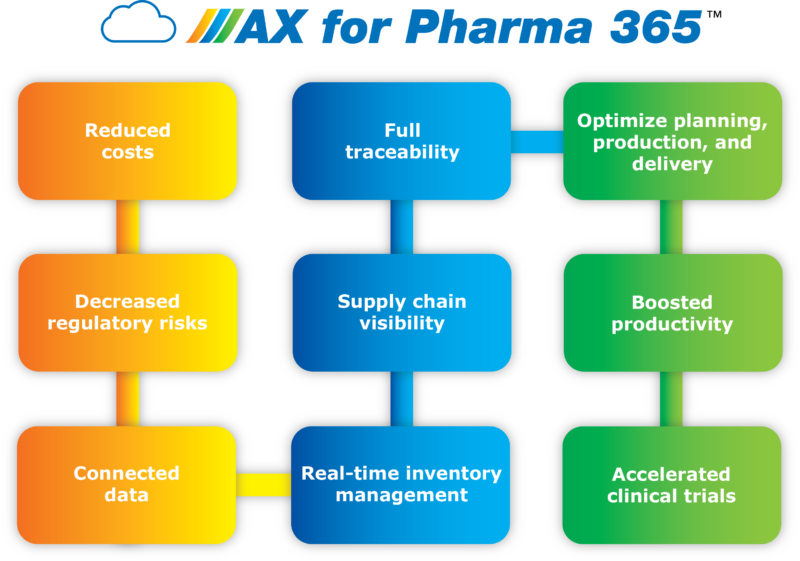
Unlike generic enterprise resource planning (ERP) solutions, the AX for Pharma 365 Clinical Supply™ module has been built to cope with these exact demands. It is natively integrated within Microsoft Dynamics 365, and could be interfaced with any other high-level ERP solution. This significantly improves the forecasting, planning, manufacturing, and distribution processes of clinical trial supplies — resulting in multiple benefits that will set you apart from the competition.
By increasing connectivity across the entire value chain, you can realize a step change in efficiency. Not only will you be able to react to changes in demand at the touch of a button, but with predictive insights, you’ll also be able to stay one step ahead of issues that may occur during any part of the process.
On-time deliveries and real-time inventory will enable you to reduce waste. The solution also fully helps you seamlessly follow Good Manufacturing Practice guidelines and significantly reduce your exposure to regulatory risk.
The result is a whole new level of operational effectiveness that leads to faster, more effective clinical trials — with significant cost savings too.